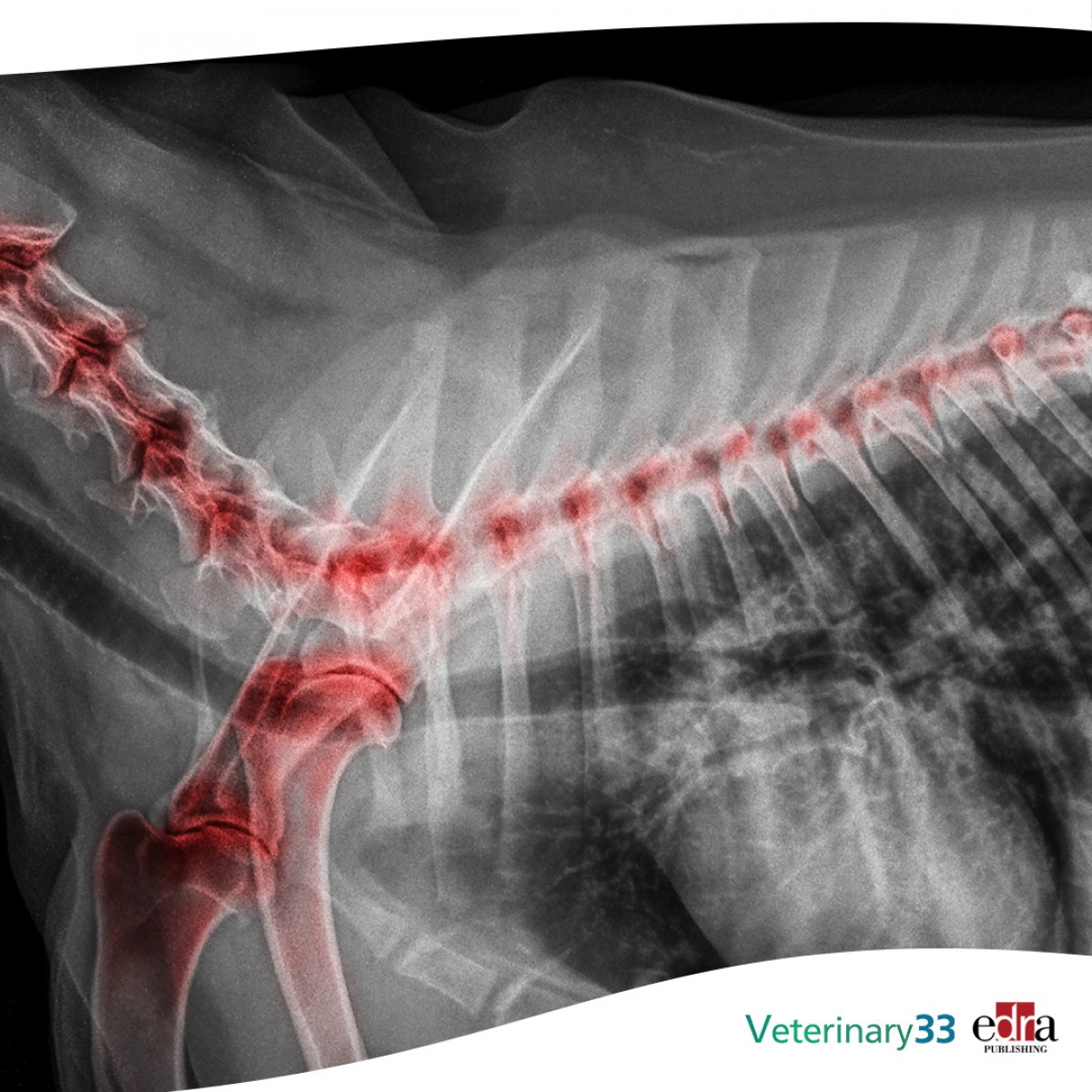
Appendicular osteosarcoma in cats
Osteosarcoma is the most common malignant primary bone cancer, but it is infrequently reported in cats. Feline appendicular osteosarcoma typically exhibits good prognosis when treated with surgery alone. A retrospective multi-institutional study was conducted to identify possible prognostic factors.
Cats diagnosed with appendicular osteosarcoma were included if initial staging and follow-up information were available. Data including signalment, tumour characteristics, treatment modalities, and survival outcomes were collected and analysed.
Fifty-six cats were enrolled; the femur was the most frequently affected bone. Eight cats had distant metastasis at admission and an additional 9 developed metastatic disease during follow-up, resulting in an overall metastatic rate of 30%. Forty-nine (87.5%) cats underwent surgery, and 4 also received adjuvant chemotherapy. Among operated cats, median time to local progression (TTLP), time to distant progression and tumour-specific survival (TSS) were not reached. One- and 2-year survival rates were 66% and 55%, respectively. Seven (12.5%) cats received no treatment; 1- and 2-year survival rates were 25% and 0%, respectively. Operated cats had significantly longer TTLP (P < .001) and TSS (P = .001) compared with non-operated cats. Among operated cats, young age negatively impacted local tumour progression, while the presence of distant metastasis at diagnosis was associated with a higher risk of tumour-related death.
This study reaffirms the good prognosis for cats with appendicular osteosarcoma undergoing surgery, but sheds light on some additional factors to consider. Accurate initial staging is recommended, as the metastatic rate may exceed many previous estimations. Surgery substantially extends survival time, whereas the role of chemotherapy remains uncertain.
“A retrospective Italian Society of Veterinary Oncology (SIONCOV) study of 56 cats with appendicular osteosarcoma”. Laura Marconato, et al. Vet Comp Oncol. 2024 Feb 7. doi: 10.1111/vco.12966.
Source: https://onlinelibrary.wiley.com/doi/full/10.1111/vco.12966












List
Add
Please enter a comment