Zoetis is Redefining the Feline Veterinary Experience with the Commercial Launch of Bonqat® (pregabalin oral solution) in the U.S.
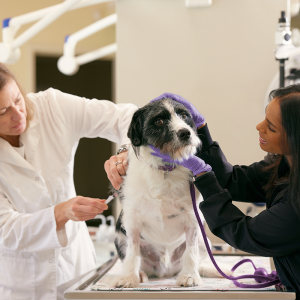
Zoetis today announced the U.S. commercial launch of Bonqat® (pregabalin oral solution) to veterinary clinics throughout the U.S. The availability of Bonqat, which Zoetis will market and distribute under an exclusive agreement with Orion Corporation, builds on the extensive portfolio of Zoetis products dedicated to advancing feline health. Bonqat was approved by the U.S. Food and Drug Administration (FDA) on November 17, 2023, for alleviation of feline acute anxiety and fear associated with transportation and veterinary visits, which can be a barrier to routine care. Prescribed by a veterinarian, Bonqat is a liquid medicine administered by mouth to cats at home by pet owners.
“Maintaining routine checkups is essential to cats’ overall health, yet only 40% of cats visit the veterinarian annually, compared to 82% of dogs.1 The stress of veterinary visits on cats and their owners can prevent cats from receiving the care they need,” said Dr. Tara Bidgood, Vice President, Veterinary Professional Services & Medical Affairs, U.S. Petcare at Zoetis. “Bonqat offers a unique solution to cat care because it not only makes it easier for pet owners to get cats to their veterinarian but also serves as a catalyst for cats to receive more comprehensive care once at the clinic, including diagnostics and preventative care for fleas, parasites and vaccinations. We’re proud to bring Bonqat to the U.S. to continue supporting the feline health journey and contributing to cats’ overall health and wellbeing.”
Feline Acute Anxiety and Fear and Its Impact on Feline Health
Many cats suffer from anxiety and fear related to travel and veterinary visits, with 59% and 66% of cats reported to exhibit signs of distress during car travel and during veterinary visits, respectively.2 Common signs of acute anxiety and fear can range from subtle to severe and include freezing, hiding, vocalizing, tail flicking, increased breathing and panting, restlessness, hissing, scratching and biting.3
If untreated, acute anxiety and fear may worsen with additional experiences, compounding the distress of being placed into a carrier and being transported to and from the clinic and reducing the chance of receiving a comprehensive physical exam,4 which can impact a cat’s lifelong wellbeing.
“Feline anxiety doesn’t just impact cats – in fact, more than half (57%) of cat owners reported feeling anxious when preparing for veterinary visits and associated travel5 because of their cat’s stress,” said Margaret E. Gruen, DVM, MVPH, PhD, DACVB, Associate Professor of Behavioral Medicine at North Carolina State University. “Removing these barriers can help cultivate a more positive and productive veterinary experience for both cats and their owners by focusing on cats’ general well-being rather than the anxiety of the visit itself, which can also help maintain the unique human-animal bond.”
Bonqat is a targeted solution formulated to meet the unique needs of cats with small dosing, slow elimination time and easy administration by simply mixing the medicine with food or administering directly into the cat’s mouth.
About Bonqat (pregabalin oral solution)
Bonqat is not a sedative.* It’s a targeted solution that directly impacts the neurotransmitters (brain chemicals) involved in feline acute anxiety and fear associated with transportation and veterinary visits, helping to create the proper emotional balance needed to support the visit. Bonqat is administered approximately 1.5 hours before transport and is slowly eliminated, allowing flexible timing for transport, a thorough exam, and the ability to extend a cat’s stay if diagnostic tests or further evaluations are needed.
*In a clinical study, 3 of 108 cats experienced sedation that resolved without treatment.6
Important Safety Information
Not for human use. Do not use in cats with known hypersensitivity to pregabalin. Use with caution in cats with cardiac disease, hypertension, renal disease, or severe systemic disorders. The safe use of BONQAT has not been evaluated in breeding, pregnant, or lactating cats; in cats younger than 7 months of age; in cats with cardiac disease, hypertension, renal disease, or severe systemic disorders; or in conjunction with opioids and other sedatives. The most common side effects are incoordination, lethargy, and vomiting. See full Prescribing Information at BonqatPI.comThis links to a pdf file.
Indications
BONQAT is indicated for alleviation of acute anxiety and fear associated with transportation and veterinary visits in cats.
About Zoetis
As the world’s leading animal health company, Zoetis is driven by a singular purpose: to nurture our world and humankind by advancing care for animals. After innovating ways to predict, prevent, detect, and treat animal illness for more than 70 years, Zoetis continues to stand by those raising and caring for animals worldwide – from veterinarians and pet owners to livestock farmers and ranchers. The company’s leading portfolio and pipeline of medicines, vaccines, diagnostics and technologies make a difference in over 100 countries. A Fortune 500 company, Zoetis generated revenue of $8.5 billion in 2023 with approximately 14,100 employees.
Source: www.zoetis.com













List
Add
Please enter a comment