Researching Feline Idiopathic Cystitis
by Jenny Alonge
Researchers at Michigan State University College of Veterinary Medicine (MSU-CVM) are currently enrolling cats affected by chronic idiopathic cystitis to evaluate a potential new and novel treatment. They are investigating a cell-based therapy that uses mesenchymal stem cells (MSC) derived from the patient’s fat tissues.
Feline idiopathic cystitis
Feline idiopathic cystitis (FIC) is a bladder disorder that accounts for about two-thirds of feline lower urinary tract disease (FLUTD) cases that affect a quarter to a half million cats annually in North America. By definition, FIC is a disorder of unknown cause, but several abnormalities that are common in affected cats may contribute to the disease. These include:
- Defective bladder lining — The bladder lining is composed of polysulfated glycosaminoglycans (PSGAGs), which insulates the bladder tissue from urine. In FIC cats, this PSGAG lining is defective and deficient, possibly allowing damage to the bladder’s transitional epithelial cells, which would cause irritation and inflammation.
- Neurogenic inflammation — Bladder nerves may be stimulated by the brain in response to stress, causing a neurotransmitter release that exacerbates local pain and inflammation.
- Stress — FIC is frequently triggered by a stressful event.
- Abnormal stress response — FIC cats appear to respond abnormally to stress. Normal cats release catecholamines and cortisol in response to stress, but FIC cats have high catecholamine levels and below normal cortisol levels.
FIC, which is the most common bladder disorder affecting young to middle aged cats, is characterized by frequent, bloody, and painful urinations, and house soiling, and life-threatening urinary obstructions in severe cases. Regardless of treatment, most cats’ signs subside in about a week, although some cats experience ongoing issues or frequent sign recurrence, and their disease is classified as chronic idiopathic cystitis. No specific test exists to detect FIC, and the condition is diagnosed by excluding other disorders, such as bladder stones, bacterial infections, bladder tumors, and behavioral disorders, that cause similar signs. A thorough work-up involving urinalysis, bacterial culture, and urinary tract imaging using X-ray studies, ultrasound, and urethrocystoscopy must be performed. A FIC diagnosis can be made only by ruling out other recognized FLUTD causes. Despite decades of research, no treatment has been found that can consistently and effectively manage cats affected by chronic idiopathic cystitis. Current management techniques include using antispasmodics, tranquilizers, and analgesics to manage the pain and urinary discomfort, changing the cat’s diet, minimizing stress, and providing environmental enrichment.
Mesenchymal stem cell research project
Adult mesenchymal stem cells are self-renewing progenitor cells that can differentiate into more specific cell types. These cells possess incredible immunomodulatory properties that likely promote tissue healing and repair, and these characteristics have led to their use in treatments to address chronic inflammatory diseases in people and animals. Chronic inflammation is commonly seen in biopsies from chronic idiopathic cystitis cats, and many signs are likely caused by inflammatory mediators in the bladder wall.
The MSU-CVM researchers hypothesize that mesenchymal stem cell therapy will be beneficial for chronic idiopathic cystitis cats. Their goal is to investigate the efficacy of injecting autologous fat tissue-derived mesenchymal stem cells in the intraperitoneal space of cats with chronic idiopathic cystitis. In adult animals, fat tissue is one of the richest mesenchymal stem cell sources, and they plan to collect an affected cat’s fat tissue to formulate a treatment. The process entails:
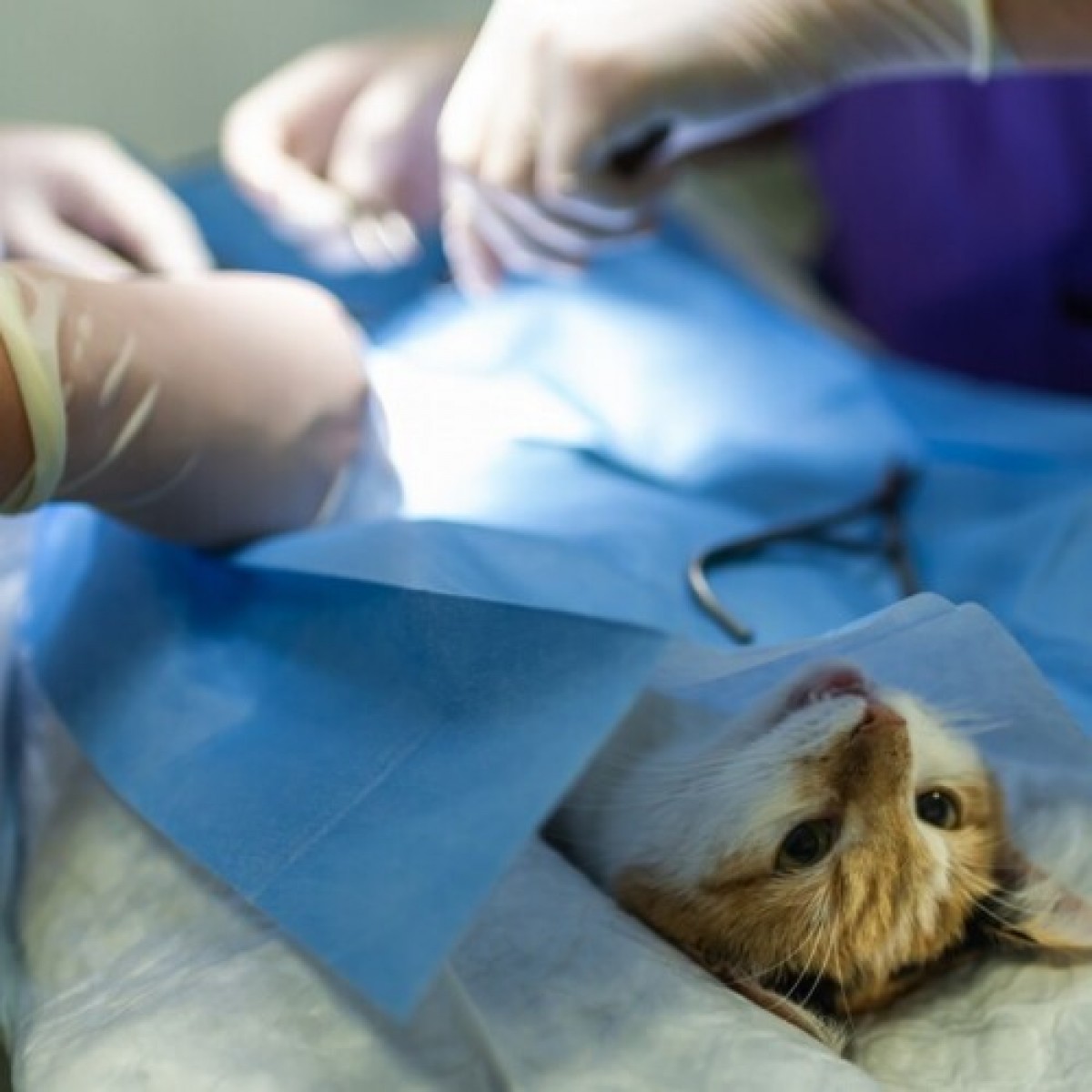
- Collecting the tissue — Cats will be placed under general anesthesia, and a small amount of fat will be collected from under their skin through a small incision.
- Preparing the sample — The mesenchymal stem cells will be purified from the fat cells and tagged with nanoparticles.
- Administering the treatment — Under general anesthesia, the mesenchymal stem cells will be injected into the cat’s abdominal cavity using ultrasound guidance to ensure the stem cells are close to the bladder and can easily reach their intended target.
- Tracking the stem cells — Magnetic resonance imaging (MRI) scans will be performed immediately before and after stem cell injection and one week after injection to track the stem cells’ migration.
The mesenchymal stem cell therapy will be considered successful based on the number and frequency of bladder signs before and after treatment. This study design is unique, because each cat is acting as their own control. Each cat’s signs will be quantified for about two months prior to stem cell injection, and then evaluated for six months following the injections to determine if each cat improves.
The researchers are recruiting indoor-only cats affected by severe, persistent, or frequently relapsing chronic idiopathic cystitis that has failed to respond to currently recommended treatment. Cat owners will be asked to fill out a detailed questionnaire about their current household enrichment practices, other household pets, and dietary and treatment history. The study costs will be covered completely, and each cat owner whose cat completes the study will receive $400.
Chronic idiopathic cystitis is a serious condition that significantly impacts an affected cat’s quality of life. Hopefully, the MSU-CVM researchers will identify a valuable treatment option to help affected cats.








List
Add
Please enter a comment